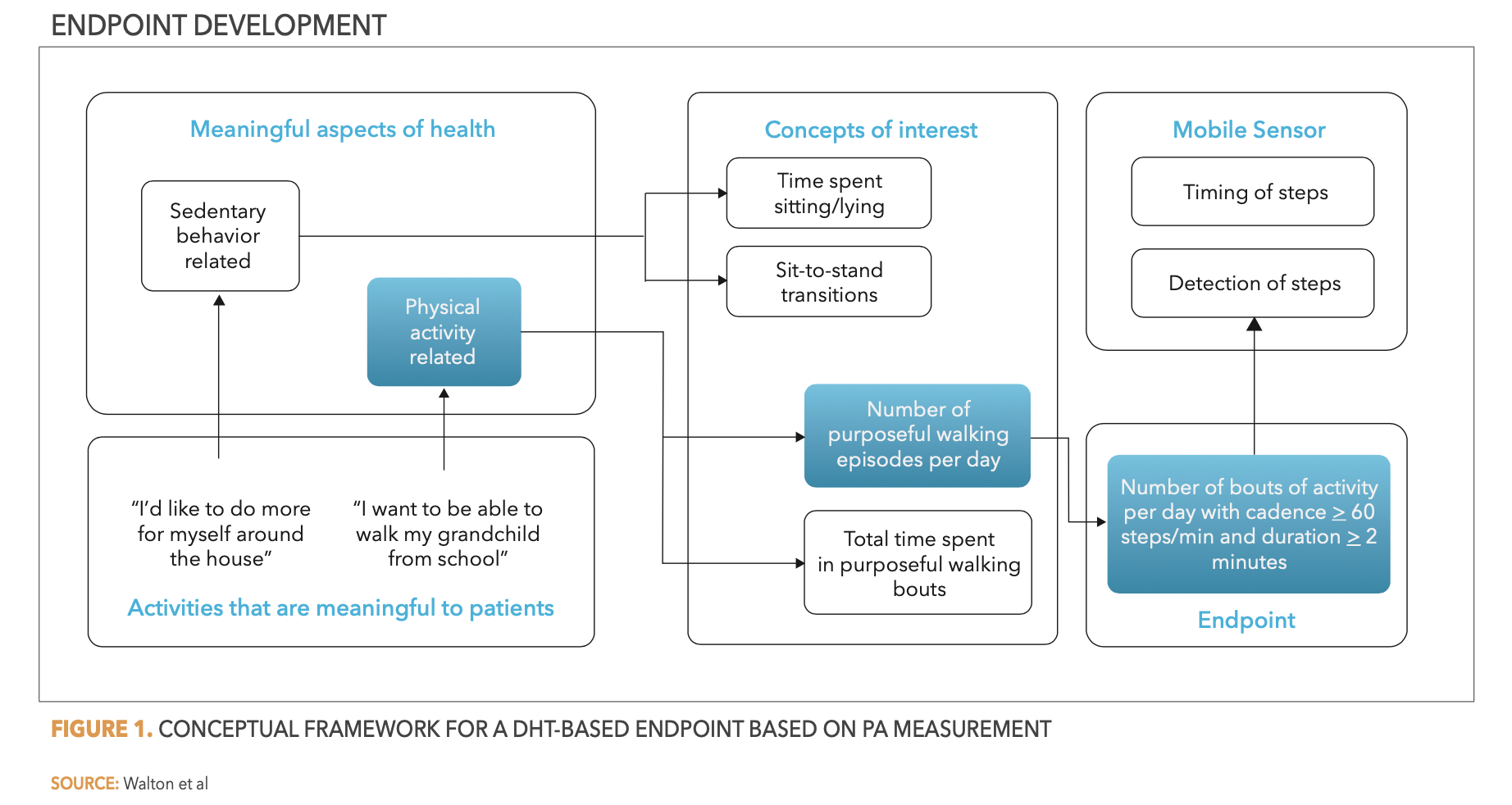
Digital Progression Biomarkers as Novel Endpoints in Clinical Trials: A Multistakeholder Perspective - IOS Press

Development of Novel, Value-Based, Digital Endpoints for Clinical Trials: A Structured Approach Toward Fit-for-Purpose Validation | Pharmacological Reviews

Development of Novel, Value-Based, Digital Endpoints for Clinical Trials: A Structured Approach Toward Fit-for-Purpose Validation | Pharmacological Reviews

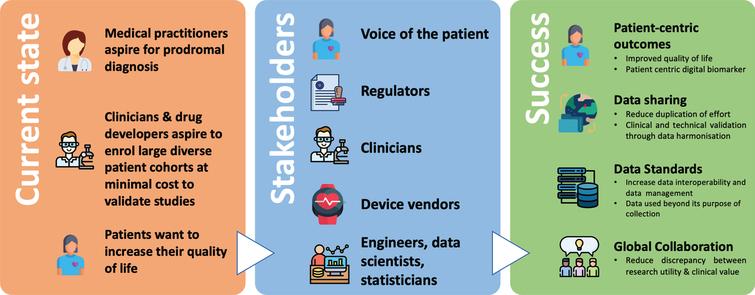
Advancing Digital Medicine to Optimize Human Health: 2019 in Review – Digital Medicine Society (DiMe)

PDF) Development of Novel, Value-Based, Digital Endpoints for Clinical Trials: A Structured Approach Toward Fit-for-Purpose Validation

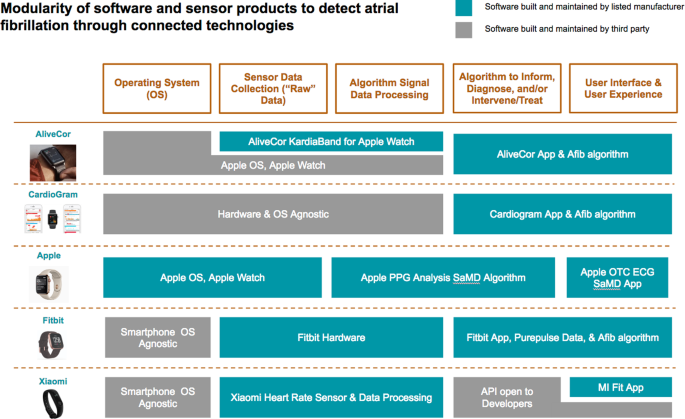
Digital endpoints: Global opportunities and clinical, data protection and other challenges - MedCity News

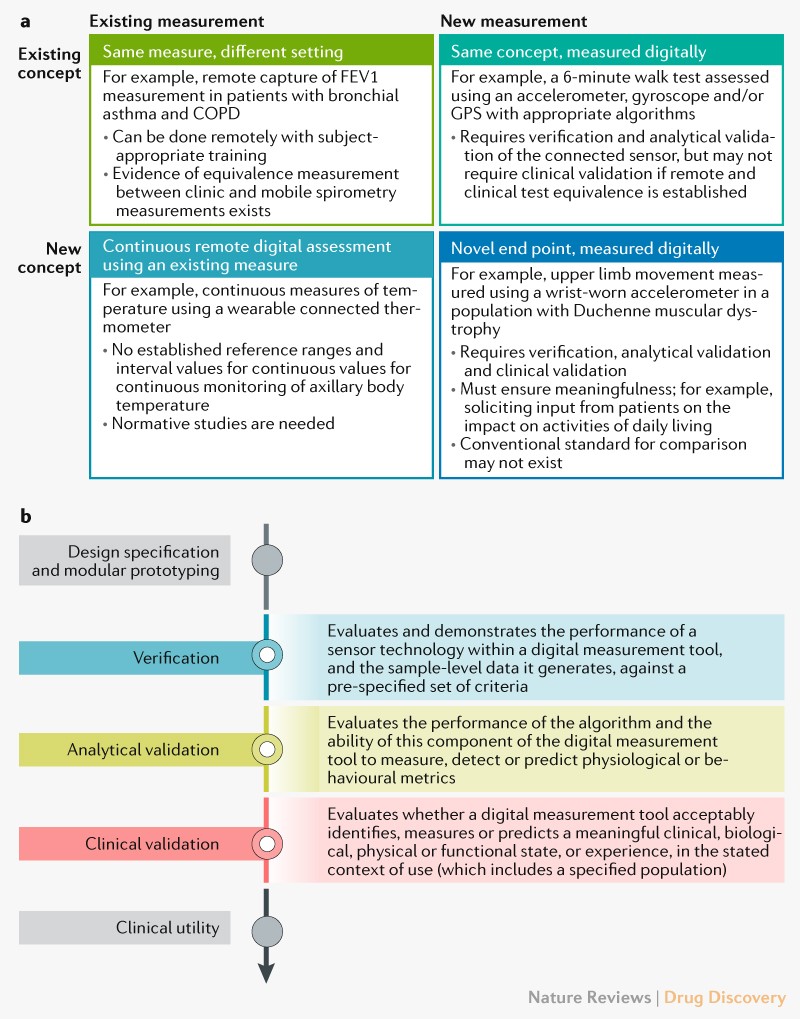
Using Digital Health Technology to Better Generate Evidence and Deliver Evidence-Based Care - ScienceDirect




![Essential Steps to FDA Approval for Digital Biomarkers [Infographic] Essential Steps to FDA Approval for Digital Biomarkers [Infographic]](https://www.vivosense.com/hubfs/images/blog/essential-steps-to-fda-approval-for-digital-biomarkers-infographic-1260x3113px.png)