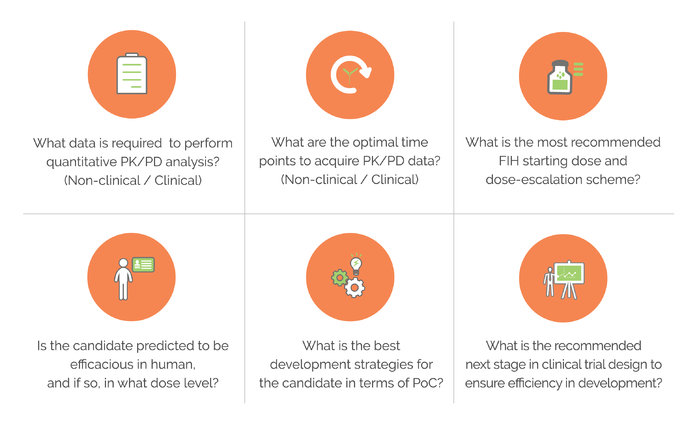
Pharmacokinetic and Statistical Considerations in First-in-Human Clinical Trials | Pharmaceutical Outsourcing - The Journal of Pharmaceutical & Biopharmaceutical Contract Services

10/20/2015 - Session II: Clinical Trial PK/PD Translated To Population Drug Use and Exposure - YouTube
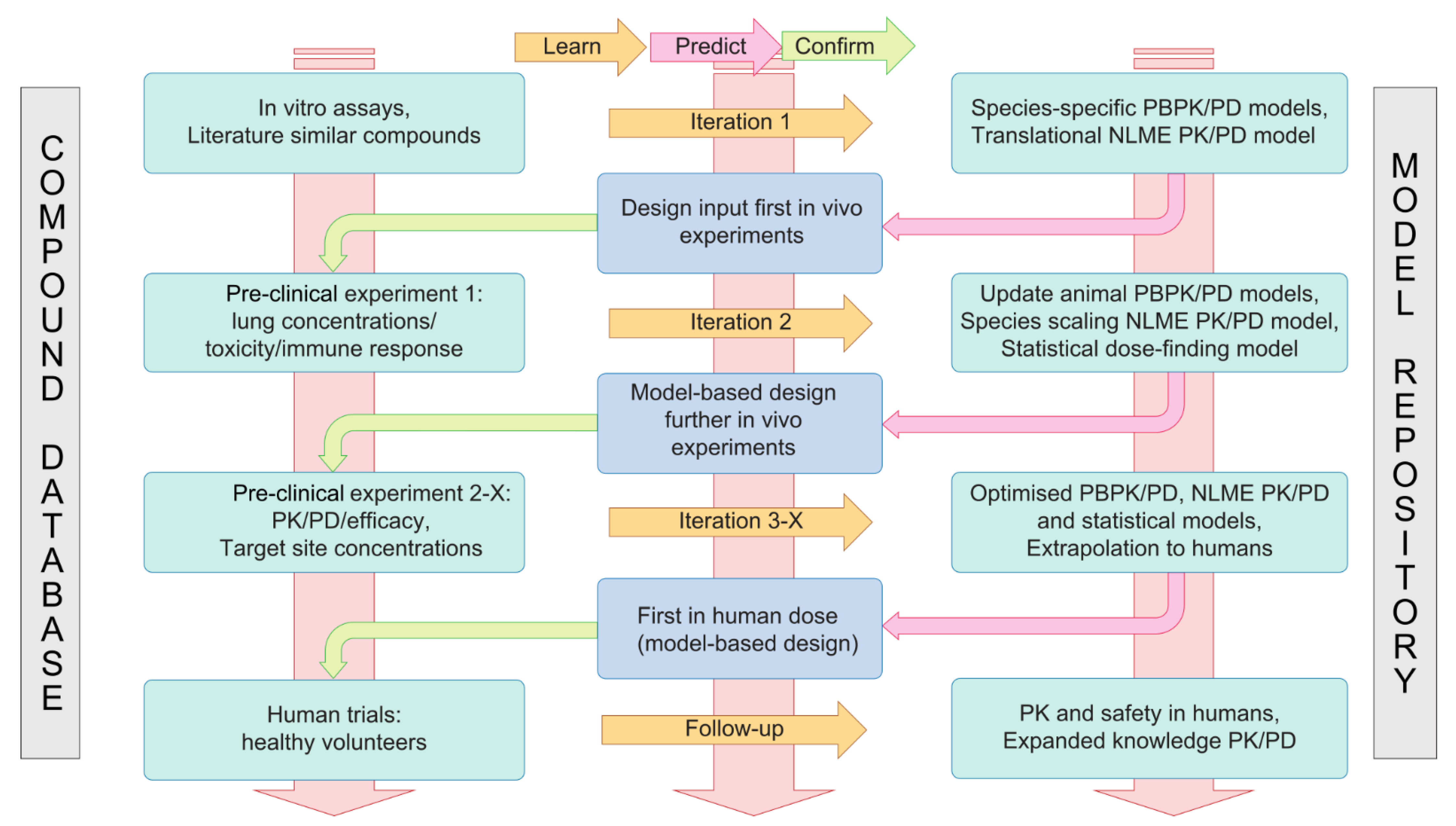
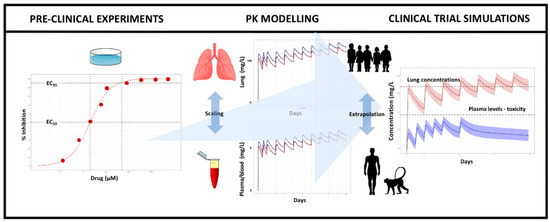
Pharmaceutics | Free Full-Text | The Use of Translational Modelling and Simulation to Develop Immunomodulatory Therapy as an Adjunct to Antibiotic Treatment in the Context of Pneumonia

Metformin Clinical DDI Study Design That Enables an Efficacy- and Safety-Based Dose Adjustment Decision | Pharmaron
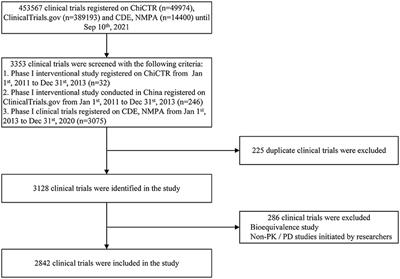
Frontiers | Trends of Phase I Clinical Trials of New Drugs in Mainland China Over the Past 10 Years (2011–2020)

On Biostatistics and Clinical Trials: Serial Blood Sample Timepoints for Comparing Pharmacokinetics Profiles Between Two Different Dose Frequencies

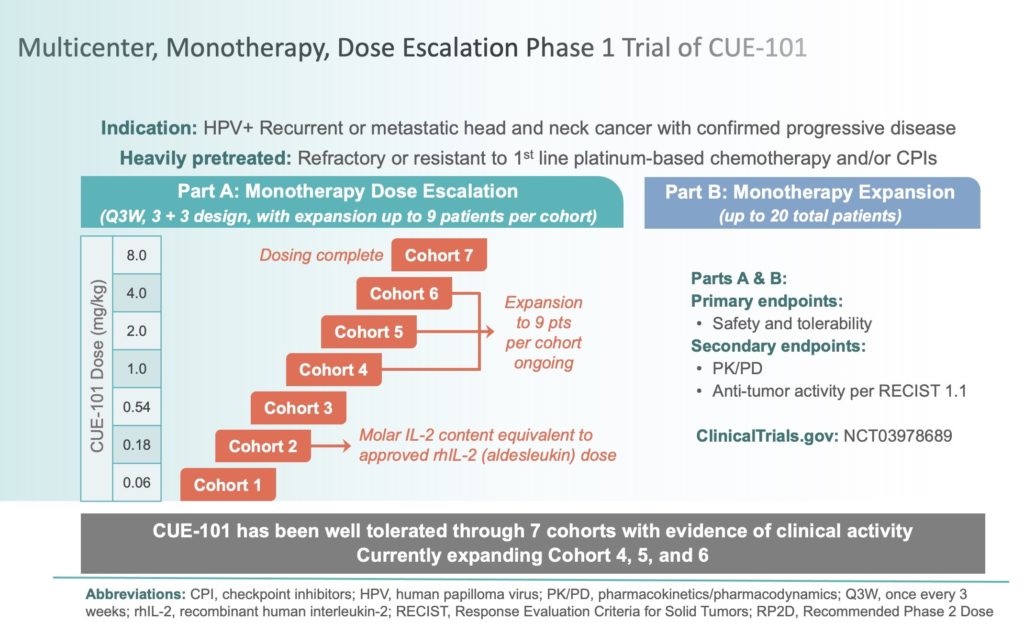
Optimal biological dose: a systematic review in cancer phase I clinical trials | BMC Cancer | Full Text
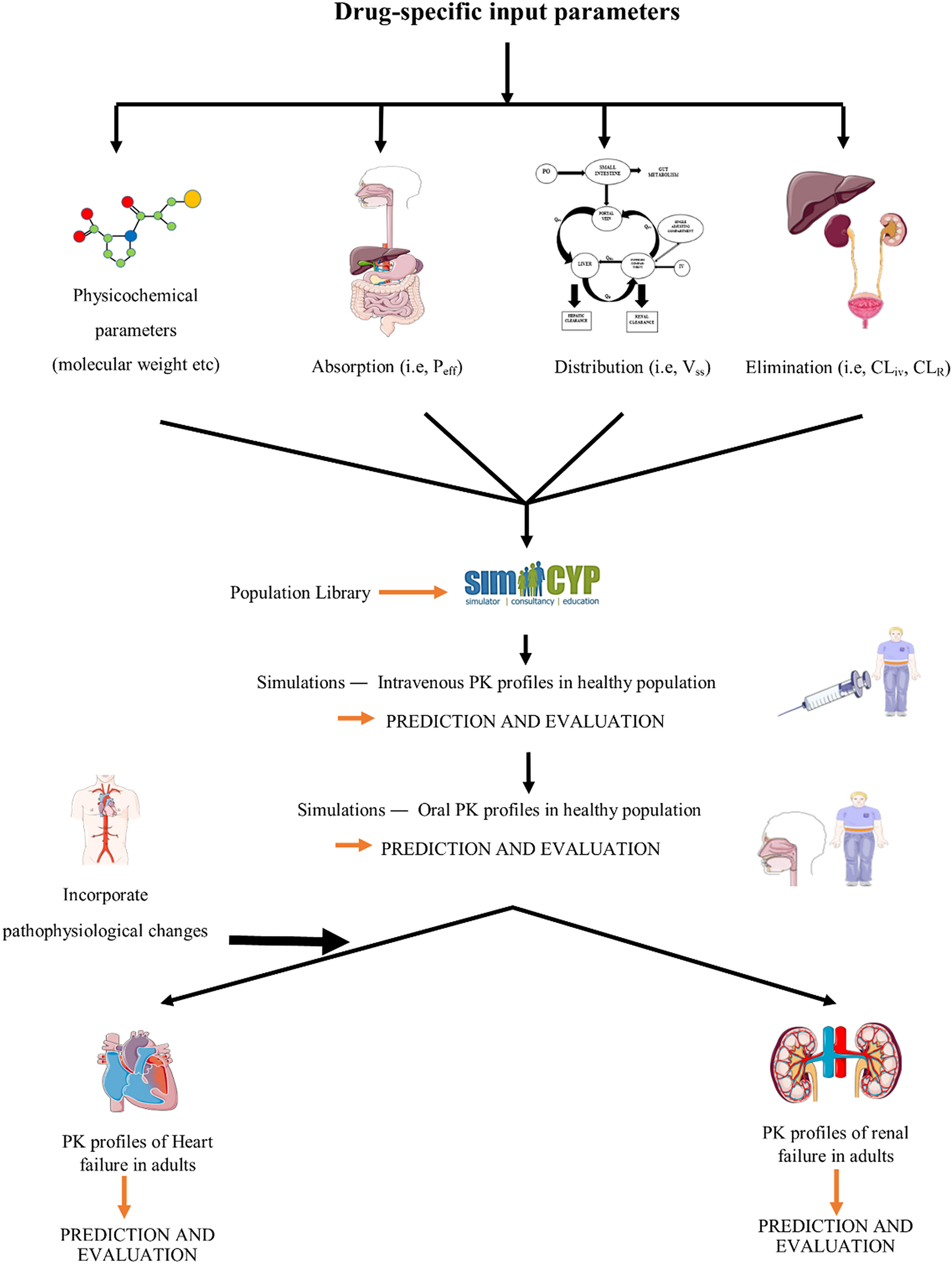
Development and evaluation of physiologically based pharmacokinetic drug-disease models for predicting captopril pharmacokinetics in chronic diseases | Scientific Reports

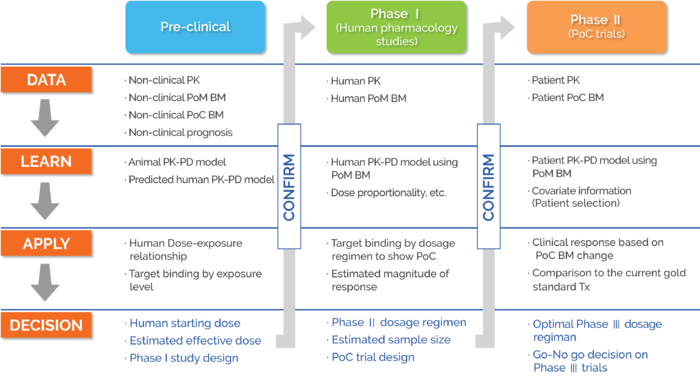
Schema of preclinical and clinical pharmacology studies important for... | Download Scientific Diagram

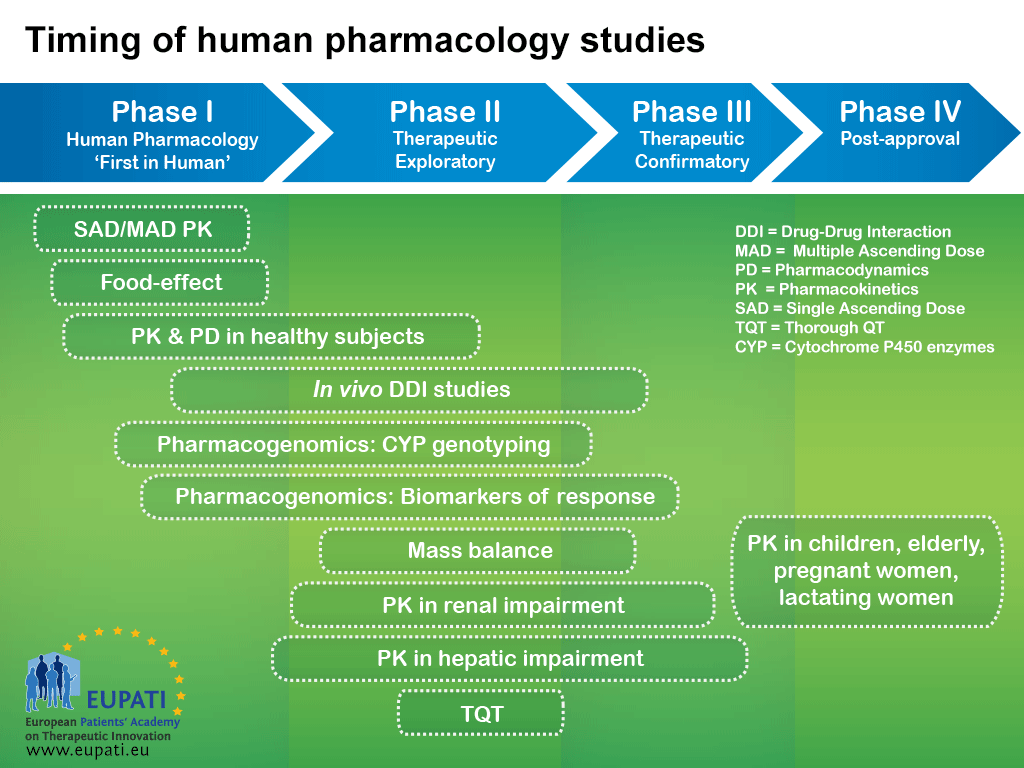
Current status and future perspective on preclinical pharmacokinetic and pharmacodynamic (PK/PD) analysis: Survey in Japan pharmaceutical manufacturers association (JPMA) - ScienceDirect






![PDF] Phase 0 Clinical Trial Strategies for the Neurosurgical Oncologist | Semantic Scholar PDF] Phase 0 Clinical Trial Strategies for the Neurosurgical Oncologist | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/ba643ff4c798a7864e6a78f009413a89e764a2c0/6-Figure2-1.png)